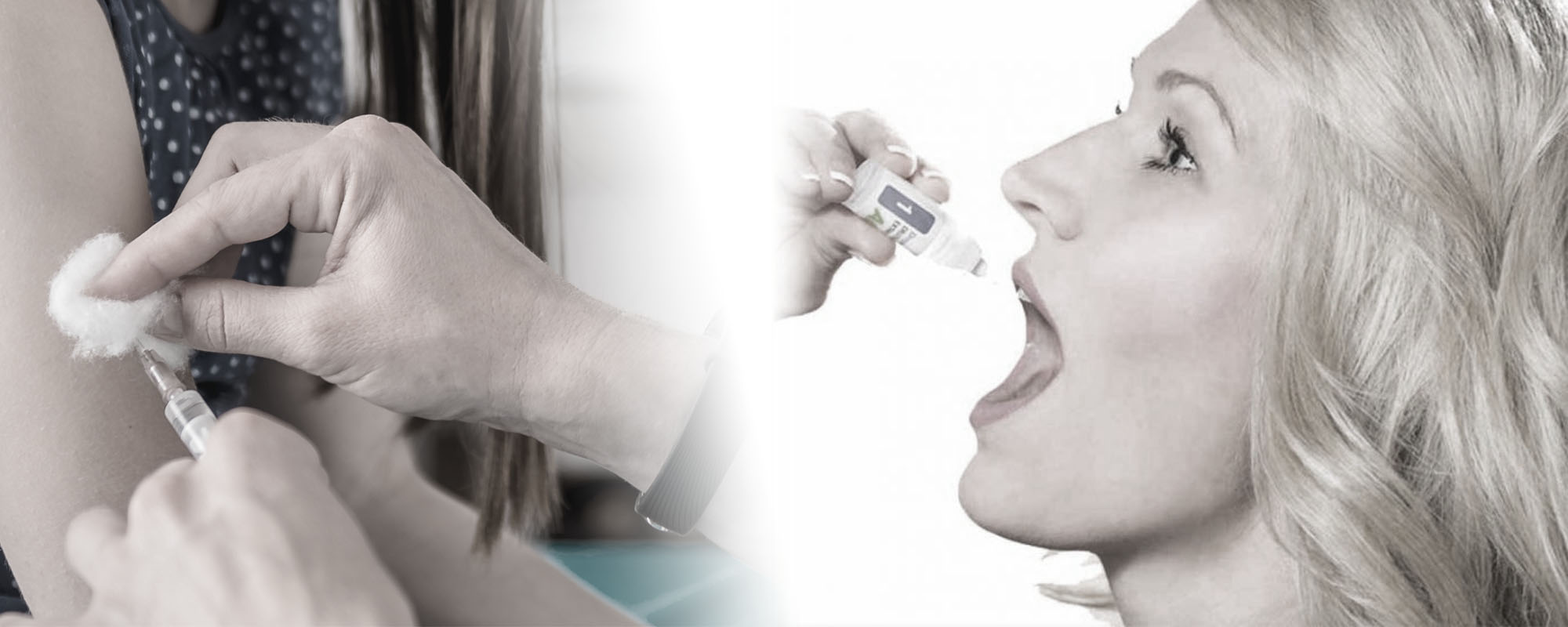
Sublingual immunotherapy for environmental and food allergies. Allergy shots for environmental allergies.
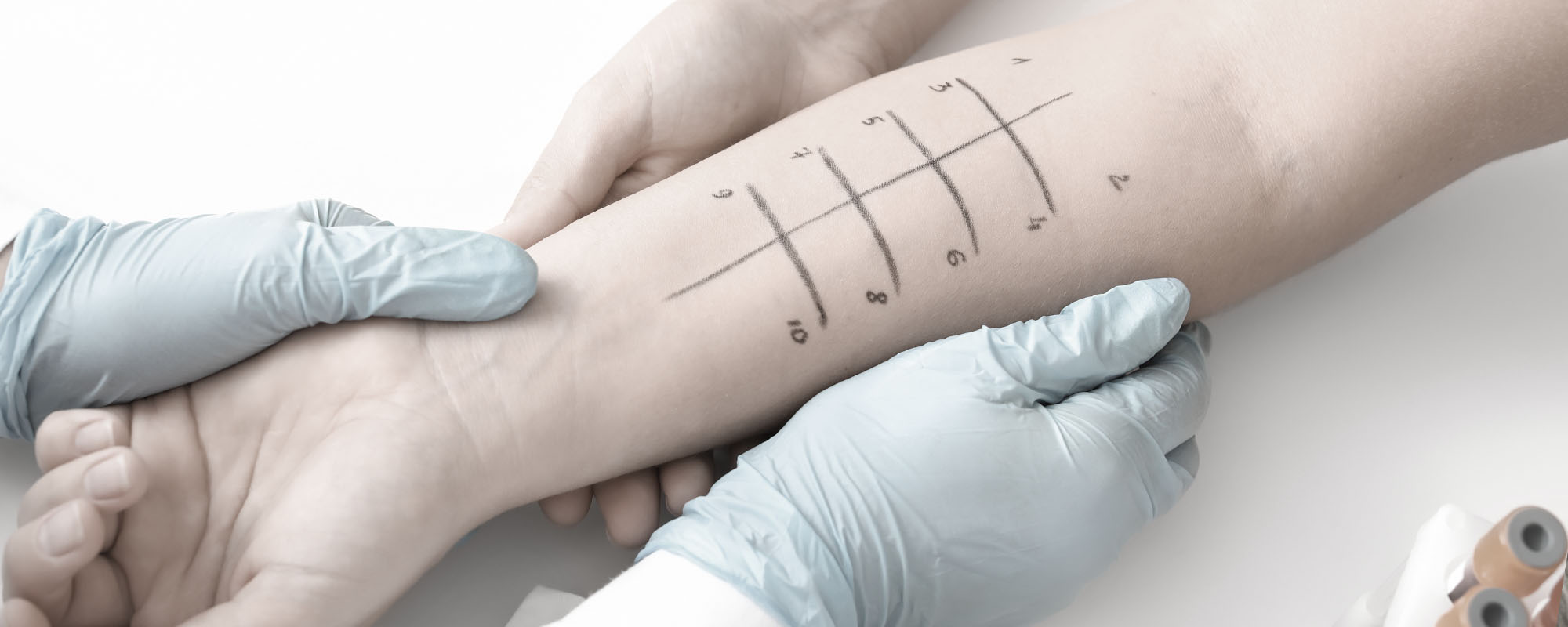
Testing for environmental allergies. Allergy Testing by Allergist & Allergy Specialist Doctors. Book Your Appointment today!

Food Allergy Testing by Food Allergy Specialists. Tree Nuts, soy gluten, honey allergies & other less common food allergies.
Medication-assisted weight management using GLP-1a, Contrave, or Phentermine.
Sublingual immunotherapy for environmental and food allergies. Allergy shots for environmental allergies.
Testing for environmental and food allergies. Food allergy testing services.
For knee & shoulder pain, hair loss, and facial skin rejuvenation.
HiFU (high intensity focused ultrasound for skin tightening and vaginal rejuvenation); Cryolypolisis to freeze fat cells away; Microdermabrasion hydra-facial, red light therapy.

Testing for gluten intolerance, gluten sensitivity, and/or testing for Celiac Disease.
Here are just a few of the benefits:
No injections.
Pain-free treatment for allergies! With sublingual immunotherapy, allergy serum is taken as drops under the tongue instead of as shots.
Take-at-home drops.
Because sublingual allergy drops are safer than shots, they can be taken at home rather than at the doctor’s office.
Safer than allergy shots.
Drops are not associated with the same degree of risk for anaphylactic reaction that shots are.
Ideal for children, too.
Children are often required to be at least 7 years old before they can start on allergy shot programs, but allergy drops have been shown to be safe and effective for children under 5.


Long-term relief.
Pills, inhalers, and nose sprays only affect the symptoms of allergy–not the allergy itself. Allergy drops treat the underlying problem, conditioning the body to stop overreacting to allergens in the environment in ways that lead to troubling symptoms.
Perfect for travelers.
Since allergy drops can be taken on the go, you can travel freely without affecting your treatment regimen. And for air travelers, the dropper bottles meet TSA standards (less than 3 ounces of liquid).
More thorough protection than standard allergy shots.
Many allergists only treat for a small selection of local pollens, but Family Allergy Clinic drops contain a mix of virtually all common allergens. (We are not aware of another allergy mix in the country that is as comprehensive as ours!) This helps ensure broad allergy protection from the full spectrum of allergens in your area and from new allergens you may encounter as you travel or move.
Natural ingredients.
Allergy drops are made up of the very things you breathe in nature and are free of the side effects of synthetic medications.
For food allergies.
Tired of having to avoid certain foods? Our food allergy drops can help you tolerate more of the foods you love.
Short and simple allergy test.
Our test consists of a brief series of superficial skin pricks. Irritation is minimal, and it lasts less than a half hour.
Contact the Family Allergy Clinic & Wellness Center 480-827-9945
*Restrictions map apply for Medicare, Medicade or any federal or state-funded programs.
Ask about specific Medicare Advantage Plans.
Not local? No problem. Our allergy specialist provides convenient services throughout Arizona and Nevada via Telehealth:

Visit Us In Person
Family Allergy Clinic & Wellness Center © | All Rights Reserved. 2024